The startle response in fish is a pattern of rapid contractions of the axial musculature used for swimming elicited by external stimuli. In zebrafish, it has long been employed for analyses of motor function, sensory physiology and basic forms of learning. This protocol measures the startle response of zebrafish larvae to vibration stimuli.
In developing zebrafish, the startle response can be quantified from day four post fertilization onwards by monitoring a larvae’s locomotor activity in response to vibratory stimulation.
The Zantiks MWP unit allows measuring the activity of multiple larvae (up to 96) and also control the duration, intensity and frequency of vibratory stimulations.
Startle response of larval zebrafish. Courtesy of Rodrigo J. De Marco, Max Planck Institute for Medical Research, Heidelberg, Germany
Experimental set up
Larval zebrafish are placed individually in each well of a 6, 12, 24, 48, or 96 well plate.
Experimental procedure
The multiwell plate with the larvae is inserted into the chamber of the Zantiks MWP unit, and the script is set up to control illumination level (complete darkness in this case), vibratory stimulation (state variables of the motor rotations causing reproducible water motions inside each well) and the locomotor activity of each larva (measured in pixels which can be converted to mm), which is written to a data file at whatever frequency is required by the experimenter – 1 second bins are often used.
Results
Video (speed, 8x) of zebrafish larvae in a 24-well plate before, during and after a single pulse of mild vibratory stimulation. The red dot in the top right hand side of the video frame depicts when the vibration occurs.
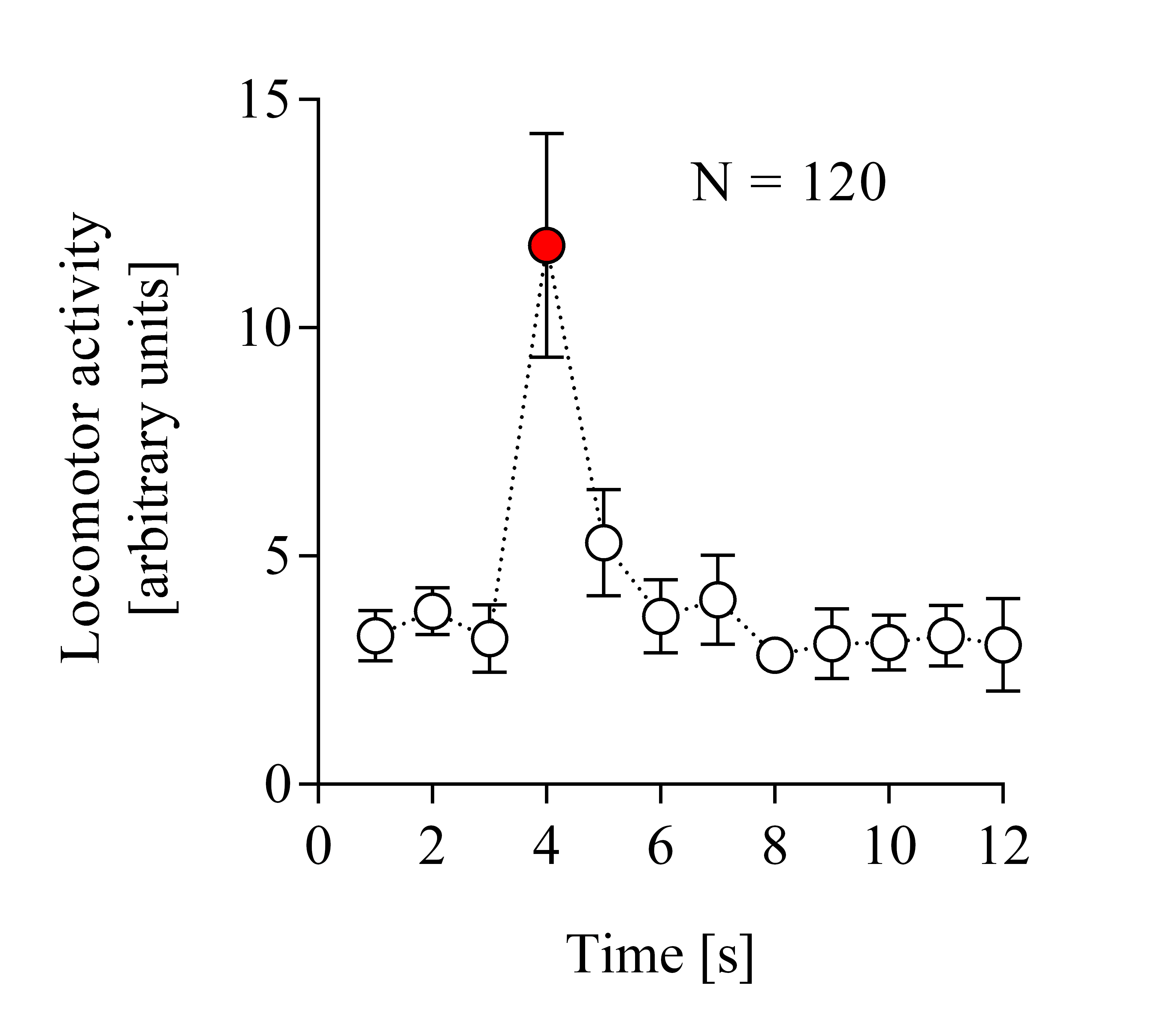
Figure depicting average locomotor activity over time in response to vibratory stimulation.
Protocol scripts download
There is one script for the startle response assay for larval zebrafish.
There will be one asset you use. This will be custom calibrated for your specific well plate or Petri dish (e.g., a 24 multiwell plate).
Assets
You will need to upload the appropriate asset into the Asset directory on your Zantiks Control Console and ensure the correct asset name is in the LOAD(ZONES,"name_of_asset") command in the script.
See the Calibrating your Zantiks unit page and Asset building in MWP unit page for details on how to create assets customised to your system.



